Oxygenated Volatile Organic Compounds (OVOCs) at L4

Left to right: Dr. Jo Dixon, Dr. Rachael Beale
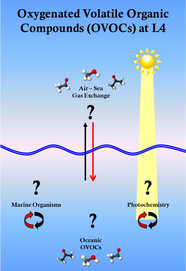
OVOC Background
Oxygenated volatile organic compounds (OVOCs) are small, reactive gases which include alcohols, aldehydes and ketones. Of particular interest to our investigations are methanol, acetaldehyde and acetone.
These species are found in the troposphere and are produced from both natural and anthropogenic processes. All three compounds are produced from industry, through vehicle emissions and are products released in the smoke plumes from those regions affected by biomass burning. Natural sources include plants and soil, in-situ atmospheric production (via oxidation of hydrocarbons) and chemical production via photolysis.

Left to right: Stephanie Sargeant, Dr. Phil Nightingale
OVOCs in the ocean are present through one of four mechanisms. (i) Through in-situ biological production; (ii) Through the action of sunlight on dissolved organic matter which can degrade higher organics to produce smaller OVOCs; (iii) Transfer from the atmosphere to the ocean or (iv) through oceanic mixing, which can transfer gases found at depth to the surface ocean. It is our aim to try and investigate which of these are the dominant pathways for OVOC production in the ocean and to assess to what extent these mechanisms are influenced by coastal seasonality.
Why study OVOCs
The atmosphere has a self-cleansing capacity which removes or reduces the abundance of pollutants via reaction with highly unstable hydroxyl radicals (HOx). Through atmospheric degradation, methanol, acetone and acetaldehyde have the ability to influence levels of HOx, thereby altering the oxidative capacity of the atmosphere. Methanol is the most abundant OVOC and its breakdown leads to the generation of formaldehyde which consequently, leads to increased levels of carbon monoxide (CO) and ozone (O3). Degradation of acetone and acetaldehyde also causes the formation of other stable trace gases such as formaldehyde, CO, carbon dioxide (CO2), O3 and peroxyacetyl nitrate (PAN). In addition to this, the photolysis of acetone can also produce acetic and peracetic acid which can contribute towards acid rain by lowering the pH of the aqueous phase in clouds.
What questions are being investigated
So, we know about atmospheric OVOCs but what about the role of the ocean in the cycling of these compounds? There are many uncertainties regarding oceanic OVOCs and our group at PML is trying to investigate the following questions at L4:
- Is there evidence that the OVOCs exhibit a seasonal cycle in concentration?
- How does the concentration of the OVOCs vary throughout the water column at L4?
- Do marine heterotrophic bacteria consume OVOCs for energy and growth, and if so, at what rates? Does seasonality affect these rates?
- Can we unravel the dominant process for producing and destroying OVOCs in the surface ocean; Photochemical or biological?
- What is the likely direction of the air-sea flux at L4?
Why study OVOCs at L4?
The Western Channel Observatory (WCO) samples L4 as part of a time-series investigation. For our OVOC analyses it is the perfect platform from which to assess temporal changes in OVOC cycling. This station is subject to a pronounced seasonal cycle, with phytoplankton blooms expected in spring and to a lesser extent in autumn. The routine measurements (chlorophyll a, nutrients, temperature, salinity, community species, optics etc) that are made on a weekly basis are also critical to our analyses as they will provide the basis from which to perform correlations to try and extend our OVOC understanding. In addition to this, the locality of the station permits fast analysis after sampling which is critical to maintain sample integrity. Furthermore, the WCO enables us to investigate the first ever coastal, seasonal cycle to generate an exciting and totally novel dataset.
How do we study OVOCs?
Oceanic concentrations (performed by Dr. Rachael Beale)
There are currently few oceanic datasets available due to the difficulty in reliably extracting these reactive species from solution. The OVOCs are highly soluble, labile and are present in trace concentrations. Furthermore, analytical instrumentation is hydrophobic so removing the water and humidity is a challenging necessity! We have developed a method at PML which uses a membrane inlet in combination with a Proton Transfer Reaction / Mass Spectrometer (PTR/MS) [Beale et al., 2011]. The membrane inlet acts as an interface between the aqueous sample and a gas phase. The OVOCs present in the sample are able to pass through the membrane into the gas phase which flows directly to the PTR/MS. The PTR/MS is able to detect methanol, acetaldehyde and acetone and the production and analysis of seawater standards allows direct calibration to determine the sample concentration.
Bacterial uptake and oxidation rates (performed by Dr. Joanna Dixon & Stephanie Sargeant)
Using tracers that have been labelled with radioactive carbon e.g.14C we follow the incorporation and uptake of OVOC compounds like methanol and acetaldehyde into marine organisms and assess how much they use them as a carbon source for growth. In addition, marine organisms also use these compounds for energy generation, which we catch as 14C labelled carbon dioxide. We precipitate this as a solid carbonate so that we can quantify rates using a liquid scintillation counter.
The results so far
Recent results published by Dixon et al. (2011) have predicted that marine microbes use methanol up to a maximum rate of 8.4 nmol L-1 day-1 with in situ concentrations around 97 nM. Although kinetic experiments suggest that the in situ methanol pool at L4 during more productive months could be turned over in 7 days.
The publications
- Beale R., Dixon JL., Liss PS., Nightingale PD. (2011) Quantification of oxygenated volatile organic compounds in seawater by membrane inlet-proton transfer reaction/mass spectrometry. Analytica Chimica Acta 706: 128-134.
- Dixon JL., Beale R and Nightingale, PD. (2011) Microbial methanol uptake in northeast Atlantic waters. ISME J 5:704-716
 Western Channel Observatory
Western Channel Observatory